Mass tort cases are lawsuits that involve multiple plaintiffs from various districts. Each of these plaintiffs file lawsuits against the same entity for similar injuries. Mass tort litigation differs from a class action, which typically has a lead plaintiff filing one lawsuit on behalf of other injured people. As a result, any compensation a judge awards will be divided evenly among all class members
in a class-action lawsuit. Contrarily, mass tort cases involve numerous individual personal injury lawsuits, allowing each plaintiff to seek compensation relative to their specific injuries.
Mass tort litigation is complex and can take several years to resolve. This article provides information and updates on significant mass tort cases that are currently pending or recently completed throughout the United States.
Mass tort cases arise when corporations fail to take proper precautions to safeguard people, resulting in similar illness or bodily harm in large victim pools. Mass tort cases comprise multiple victims suing a singular or the same group of entities for negligence. The following issues typically lead to injuries observed in mass tort cases:
Exposure to toxic
chemicals
Dangerous
products
Defective
drugs
Faulty medical
equipment
Plaintiffs in mass tort cases can be victim clusters (e.g., employees of the same company, residents of the same area, patients undergoing similar medical treatments) or consumers of a similar product.
Defendants in mass tort cases are generally large corporations, such as pharmaceutical companies and consumer product manufacturers. Since mass tort cases can involve thousands of plaintiffs, collecting evidence to accurately represent the total interests of each plaintiff can be challenging.
Often, multiple attorneys and law firms handle cases for varying plaintiffs within the mass tort litigation. With the various plaintiffs, attorneys, and law firms, case management can get complicated. Cases can take several years to settle or go to trial. Therefore, case management is critical in ongoing mass tort litigation to ensure plaintiffs receive the highest quality of service and consistency.
Major ongoing or recently settled mass tort litigation in the U.S. includes:
Elmiron is a bladder pain medication used to treat Interstitial Cystitis (IC), a chronic bladder condition. Recent research linked the drug to degenerative eye disease, leading injured victims to file a series of lawsuits against Janssen Pharmaceuticals, a subsidiary of Johnson & Johnson.
The lawsuits allege that Janssen Pharmaceuticals failed to warn users of the dangerous side effects of Elmiron, thereby knowingly putting consumers at risk of injury.
In 2019, Kaiser Permanente, Emory Eye Center, and Harvard conducted research and clinical studies that showed Elmiron usage could cause maculopathy, resulting in a loss
of central vision.
In line with their research, Kaiser Permanente studies found that 24% of 91 Elmiron users had pigment damage causing poor vision after fifteen years of using Elmiron.
They also discovered a 42% higher toxicity rate in patients that took 1500 mg or more of Elmiron. In addition, Emory Eye Center concluded that Elmiron caused significant eye degeneration and vision loss after seven years of usage, and Harvard research showed severe eye degeneration even in users that had stopped taking Elmiron.
The Elmiron (Pentosan Polysulfate Sodium) Products Liability litigation is currently pending, centralized in the United States District Court District of New Jersey.
Three lawsuits filed against Janssen Pharmaceuticals include:
Kimberly Pelczar was the first to file a lawsuit on March 26, 2020, filing the complaint in the U.S. Court for the District of Connecticut. She alleged the defendants misled people who used Elmiron, and the medical community in general, by failing to disclose that the long-term use of Elmiron could lead to retinal damage.
She began using Elmiron in 2015 to treat IC and claims she also began to experience vision problems that same year. She underwent multiple examinations for her vision problems from 2016 to 2019, and in 2019, she received a diagnosis of “permanent retinal injury and vision loss due to Elmiron toxicity.
Barbara English filed a lawsuit in the U.S. District Court for the Eastern District of Pennsylvania on May 24, 2020, alleging the defendants failed to exercise ordinary care in the manufacture, sale, labeling, and marketing of Elmiron, thereby willfully thwarting users’ knowledge of the drug’s high risk of unreasonable harm.
She began taking Elmiron in 2001 for bladder problems and started experiencing vision problems in 2017. By 2019, an eye specialist diagnosed her with “permanent retinal injury and vision loss associated with Elmiron.”
Tina Pisco filed a lawsuit in the U.S. District Court for the Eastern District of Pennsylvania on May 24, 2020, alleging the defendants’ negligence caused various injuries and vision loss. She began taking Elmiron in 2012 for bladder pain and, in 2018, received a diagnosis of “permanent retinal injury and vision loss associated with Elmiron.”
The Judicial Panel on Multi-District Litigation (JPMDL) created an MDL on December 15, 2020, in the District of New Jersey for Elmiron lawsuits. The manufacturers and distributors are the main defendants, consisting of the following subsidiaries and parent companies:
The judge overseeing the MDL has held three status conferences thus far on January 8, 2021, February 10, 2021, and March 10, 2021, and will continue to keep a status conference every 30-45 days. These regular hearings allow both sides to provide updates on the status of discovery or evidence collection and address issues as needed to keep the litigation progressing smoothly and timely.
As of August of 2022, the United States District Court of New Jersey has been holding Case Management Conferences approximately twice per month as cases continue to be added to the MDL. As of July 13, 2022, there were 1,149 cases in the federal MDL in addition to 169 state cases, an increase by more than 200 federal cases and 43 state cases since early June.
The parties have selected the following bellwether trials from an initial pool of 20:
Thousands of active and retired U.S. Army service members filed lawsuits against the manufacturing company 3M for creating and marketing defective earplugs resulting in auditory injuries. The service members alleged the company knew the earplugs were faulty as early as 2000 but failed to take any action. Injured persons filed 139,000 individual claims in the Northern District Court of Florida.
The main defendant named in the lawsuits is industrial manufacturer Aearo Technologies, acquired by 3M in 2008. The Combat Arms earplugs (CAEv2) became standard issue earplugs for all U.S. military service members around 2003. However, the public remained in the dark about the defect found within the earplugs until a $9.1 million settlement in 2018 with the Department of Defense.
The mass distribution and use of the CAEv2 earplugs aimed to reduce auditory injuries. Still, during the earplugs’ use, between 2001 and 2006, disability payments for hearing loss and tinnitus (ringing in the ears) increased by 319%. By 2007, approximately 52% of service members suffered severe hearing loss.
The company marketed the earplugs as dual-ended, allowing users to hear surrounding sounds while protecting them from loud noises. Unfortunately, the stems were too short, and they couldn’t fit properly in the ear canal. This defect rendered the earplugs ineffective at providing proper noise protection.
Internal records showed that the earplugs were never appropriately tested, and subsequent trials ended abruptly after testing only eight subjects under skewed conditions. Further investigations also revealed that the company admitted knowing the earplugs were defective and advised that user instructions be revised. Yet, they made no efforts to follow through with those revisions.
By April 2020, over 139,000 claims by 11,000 plaintiffs were pending and consolidated into a mass tort case in the Northern District Court of Florida. By June 2021, 3M faced more than 230,000 lawsuits, handing this litigation the title of “the largest consolidated federal mass tort in U.S. history.”
In March 2021, the court chose three separate cases to move forward as bellwether trials or essentially test cases intended to see how a trial might go concerning a widely contested issue. These trials can often steer settlement discussions, depending on the outcomes.
The three lawsuits chosen to be a part of the first series of bellwether trials included:
On June 21, 2021, Reuters reported that the third bellwether trial resulted in a total compensation award of $1.7 million against 3M for their part in injuries sustained by a U.S. Army veteran suffering from tinnitus. A reduction in the final payout is customary since the Pensacola, Florida jury found 3M was only 62% liable for the plaintiff’s injuries due to its failure to provide adequate warnings about its product.
However, this means the jury found the plaintiff 38% liable, rejecting claims that the product was defective. Based on these findings, 3M is looking into a possible appeal.
This latest ruling is the second loss for 3M, although it received a victory in the second bellwether trial in May 2021.

As of June of 2022, the court had concluded 16 bellwether trials, and 233,883 cases were pending, according to a Law360 report. Upon the determination that the litigants had significant information necessary for a resolution, the court ordered the parties into mediation. Without mediation, the remaining cases would be shifted to the nation’s 94 federal districts with an average of 25 cases per district, placing a heavy burden on the courts.
The Washington Post reported that of the 16 bellwether cases that were heard, 10 cases were decided in favor of the defendants. Overall, plaintiffs won a total of $77.5 million in damages, with the largest judgment of $77.5million awarded in the final bellwether case. 3M has announced intentions to appeal these verdicts.
As of July of 2022, 3M has also announced that it will file a Chapter 11 bankruptcy, claiming that such a move would be the most equitable and efficient means of resolving the massive number of remaining claims. The manufacturer has pledged to initiate a $1 billion trust fund for the benefit of victims.
The brand name for the drug ranitidine is Zantac, a heartburn medication available via prescription and non-prescription forms and used widely. The Food and Drug Administration (FDA) announced a recall of the drug in April 2020 after discovering it contained carcinogens or cancer-causing substances.
Estimates show that more than 15 million Americans were taking Zantac at the time of the recall, and millions more were taking the medication in its generic versions. Valisure, a Connecticut-based online pharmacy, first discovered the dangerous side effects of Zantac in September 2019 and informed the FDA of alarmingly high levels of N-nitrosodimethylamine (NDMA) present in samples of the drug.
Scientists found three million nanograms of NDMA in Zantac, drastically exceeding the daily limit of 96 nanograms. As a result, the FDA asked Zantac manufacturers to send samples for testing, and significant pharmacies such as CVS, Rite-Aid, Walgreens, and Walmart suspended sales of Zantac and its generic forms.
Four plaintiffs filed the first lawsuit against Zantac manufacturers Sanofi and Boehringer Ingelheim in September 2019 in the Northern District Court of California. The plaintiffs alleged the defendants failed to warn consumers of the dangers of Zantac and that the heartburn medication had an inherent design defect that caused the drug to react negatively within the human body, creating harmful amounts of NDMA.
A second lawsuit filed in February 2020 in the District Court of New Jersey comprised over 140 claims against Zantac manufacturers. The Judicial Panel on Multidistrict Litigation consolidated the cases in the Southern District of Florida.
Most plaintiffs filing lawsuits had taken Zantac or its generic forms for several years and later developed cancer. However, some plaintiffs had not developed cancer but claimed they might develop cancer in the future, requiring medical monitoring.
The latter claims prompted the defendants to file a lawsuit to dismiss the cases, claiming that no actual physical injury had occurred in those instances and there was no proof that Zantac was ineffective. Accordingly, the defendants concluded that Zantac consumers that experienced symptom relief with no apparent adverse effects were not entitled to damages.
The plaintiffs opposed the motion to dismiss, claiming that their injuries were on a cellular level, increasing their risk of developing deadly cancer, which was counter-argued by the defendants as speculation.
As of July 2020, 670 Zantac lawsuits were pending in the MDL.
As of July 15, 2022, 2,016 cases were pending in the MDL and more than 150,000 claimants had filed injury claims against the manufacturers of Zantac. The Honorable Judge Rosenberg dismissed the cases against generic makers of ranitidine on the grounds that generic manufacturers cannot alter FDA-approved drug designs or labels.
The court has designated the following five cancers for litigation:
Bellwether case selection is well underway, and the court has set the following criteria for bellwether cases:
The bellwether trial schedule has yet to be announced.

Roundup is a weed-killing product developed in 1970 using the powerful herbicide ingredient glyphosate. By 2007, Roundup had become the most widely used weed-killing product in the U.S.
Over time, studies have linked Roundup exposure to multiple forms of cancer, such as non-Hodgkin’s lymphoma, B-cell lymphoma, and leukemia, sparking the onset of thousands of lawsuits against Roundup manufacturer Monsanto.
Plaintiff Edwin Hardeman (70) filed a lawsuit against Monsanto in 2016, alleging that his use of Roundup from the 1980s to 2015 caused his non-Hodgkin’s lymphoma and that the company withheld evidence of the popular weed-killer’s link to cancer. In March 2019, a San Francisco jury awarded the plaintiff $80 million in compensation, but an appeals court reduced the award to $25 million in July 2019.
Bayer appealed to the Supreme Court, which upheld the lower court’s ruling, leaving the $25 million verdict in place, according to the Washington Post. The Supreme Court also ruled against Bayer
in a second ruling, upholding an $87 million verdict in favor of Alva and Alberta Pilliod of California, which included $70 billion in punitive damages, according to Bloomberg News.
The Judicial Panel on Multidistrict Litigation consolidated over 3,800 lawsuits filed against Bayer (a fraction of the total 125,000 lawsuits filed) into a federal MDL in the Northern District of California in August 2020.
In June 2020, Bayer announced a $10.9 billion settlement, with approximately $8.8 to $9.6 billion allocated to settle current claims and $1.25 billion reserved for future claims.
As of August of 2022, Bayer has lost three trials that have each resolved with verdicts in the tens of millions, according to a Reuters report. In July 2021, Bayer set aside $11.6 billion for settlements and litigation, and, in anticipation of the subsequent Supreme Court ruling, set aside an additional $4.5 billion. The Bayer Corporation will remove the glyphosate version of non-professional RoundUp from U.S. store shelves by the end of 2022.
The Ninth Circuit Court of Appeals has ordered the EPA to reconsider its determination that glyphosate is not carcinogenic in response to the litigation. In a CDC study, 80 percent of U.S. organic food eaters tested positive for glyphosate in their urine.

Paraquat is another widely used weed-killing product produced by ChemChina’s Syngenta Group and Chevron USA under the brand name Gramoxone. Due to plants becoming more resistant to the familiar herbicide brand Roundup (also facing numerous lawsuits), paraquat has grown in popularity in recent years.
Unfortunately, research shows paraquat is toxic, resulting in its ban by the European Union, China, Brazil, and many other countries. In the U.S., the Environmental Protection Agency (EPA) approved paraquat only for licensed commercial use.
Studies link long-term exposure to paraquat to the increased risk of developing Parkinson’s disease. This degenerative nervous system disorder leads to loss of muscle coordination and causes mild to severe disability.
Injured persons filed multiple product liability lawsuits against Syngenta Group and Chevron, alleging design defects and failure to warn consumers of paraquat’s adverse health effects.
Chevron’s defense team claimed that it had not sold paraquat in 35 years and vehemently denied its role in patients’ injuries, primarily the development of Parkinson’s disease.
On June 7, 2021, the Judicial Panel on Multidistrict Litigation consolidated numerous lawsuits into an MDL in the Southern District of Illinois. As of June 22, 2021, the MDL saw an increase of 116 lawsuits, bumping the number of pending lawsuits to 173. In addition, 10 related cases are pending in other federal courts, and 31 lawsuits are pending in state courts.
The presiding judge tentatively scheduled the first trial to commence in November 2022.
As of July 15, 2022, there were 1,448 pending cases in the Paraquat MDL. In February 2022, Chief Justice Nancy Rosenstengal dismissed the plaintiffs’ public nuisance and consumer protection claims but allowed the defective product and failure to warn claims to move forward.
The defense had also moved to dismiss all claims in some states based on those states’ respective statutes of repose, but the court agreed with the plaintiffs’ assertion that these limitations could be extended in the face of evidence of overly fraudulent acts by the defendants to conceal information about the product’s dangers, as exists in the case of Paraquat.
The court initially chose 16 cases for discovery and has narrowed them to the following six cases for case-specific discovery:
The first three bellwether trials were initially scheduled for November 15, 2022, June 12, 2023, and September 18, 2023. However, the discovery process has been extensive, and the discovery schedule has been amended three times. In the latest amended schedule, the deadline for the final depositions has been extended to February 2023. The court has also indicated that it does not intend to further reduce the number of bellwether cases.
Hernia meshes are medical devices used to protect damaged tissue after hernia surgery. The mesh attaches with stitches, staples, or glue and the pores in the mesh allow the tissue to grow into the device and accelerate recovery. Hernia mesh is used in 90% of hernia surgeries annually in the United States.
The USDA discovered hernia mesh complications, including pain, infection, bowel obstruction, organ perforation, and hernia recurrence. Some of these complications require patients to undergo additional surgeries to remove the hernia mesh. In addition, some damages are permanent, resulting in long-term health issues or chronic pain.

Thousands of plaintiffs filed lawsuits against hernia mesh manufacturer Ethicon Inc, a subsidiary of Johnson & Johnson. The plaintiffs alleged serious health problems associated with their hernia meshes, prompting a voluntary recall of Ethicon’s mesh products in 2016.
In 2017, a federal panel consolidated 84 lawsuits across 36 district courts against Ethicon into MDLs in the Northern District of Georgia. As of July 2021, nearly 3,600 lawsuits are pending.
The presiding judge selected four bellwether or test trials to be among the first Ethicon lawsuits to proceed to resolution. These cases are usually helpful in determining possible settlements, depending on outcomes.
The court initially scheduled the first bellwether trial (Crumbley v. Ethicon, Inc. and Johnson & Johnson, 1:18-cv-00748) to proceed on November 2, 2020, but Covid-19 resulted in widespread closures that necessitated changes to trial schedules. Due to the plaintiff’s surgery the same month of the change, its trial date of March 18, 2021, also needed rescheduling.
As a result, the trial, expected to take two to three weeks, is now set to begin on September 13, 2021, delaying the second bellwether trial (Guffey v. Ethicon, Inc. and Johnson & Johnson, 1:18-cv-00402) indefinitely and staying the scheduling for all other trial pool cases. Therefore, the court doesn’t foresee the third bellwether trial (Bovian v. Ethicon, Inc. and Johnson & Johnson, 1:17-cv-04840) moving forward until early 2022.
Two separate hernia mesh MDLs against manufacturers C.R. Bard and Atrium are also pending. C.R. Bard previously faced similar lawsuits, ending in 2011 with a $184 million payout to approximately 3,600 plaintiffs. The court set the first bellwether trial in the Atrium MDL to proceed on July 7, 2021. However, the parties removed the case from the trial docket, citing an impending dismissal.
In September 2021, Ethicon announced a confidential global settlement had been reached, which resolved all claims in the MDL that were filed by May 13, 2021. A Qualified Settlement Fund has been established and is being overseen by court-appointed Special Master Ellen K.Reisman.
The first bellwether trial against Davol and C.R. Bard in MDL 2846 also took place in September of 2021. It was a case selected by the defense, case number 2:18-cv-01509: Johns v. C.R. Bard Inc., et al. The jury found in favor of the defendants on all counts of negligence and strict liability. The plaintiff appealed the verdict on the basis of the defendants violating evidence rules and prejudicing the jury, but the appeal was denied.
The second bellwether case, Milanesi, et al. v. C.R. Bard, et al., case number 2:18-cv-01320, began in March of 2022 and concluded approximately four weeks later in favor of the plaintiffs, Antonio and Alicia Morz de Milanesi with an award of $255,000. This included a $5,000 award for loss of consortium and $250,000 in damages on the defective design claim.
The couple was not awarded punitive damages or damages for failure to warn, negligent misrepresentation, or fraudulent misrepresentation claims. The couple appealed the verdict on the grounds of inadequate damages brought on by the court’s improper instructions to the jury. The motion was denied on
July 11, 2022.
The court has selected the cases to be heard in the third and fourth bellwether trials but has not yet scheduled their commencement dates. The cases are as follows:
Cases are continuing to be filed. As of July 15, 2022, there were 17,195 pending cases in the MDL.
Medical providers started using transvaginal meshes (TVM) in the 1970s as surgical mesh to treat pelvic organ prolapse (POP), a condition in which tissues in the pelvic floor are weakened or stretched.
In 1976, the FDA gained the authority to regulate medical devices. Still, transvaginal devices were already on the market and subject to a different classification process than new medical devices, thereby classified as Class II medical devices. Class II devices don’t require premarket approval, preventing the early detection of defects and potential complications.
The FDA investigated thousands of complaints of transvaginal mesh complications between 2005 and 2010 and changed the classification of TVMs from Class II to Class III medical devices, requiring premarket approval.
Reported injuries associated with the use of transvaginal meshes due to alleged design flaws include:
Over 104,000 transvaginal mesh victims filed lawsuits against device manufacturers, including Ethicon, alleging inherent design flaws and product malfunctions causing severe side effects. Companies settled the majority of these lawsuits with an approximate $8 billion.
A Philadelphia court awarded $12.5 million to a plaintiff alleging that Ethicon’s Prolift TVM caused painful intercourse requiring surgical intervention.
A New Jersey court awarded $13.5 million to a plaintiff alleging that Ethicon’s transvaginal mesh tape caused stress urinary incontinence (SUI).
A Philadelphia court awarded $20 million and $57.1 million to two plaintiffs alleging complications from Ethicon’s TVMs. A New Jersey court awarded another $15 million to a plaintiff alleging she had to undergo corrective surgery after receiving a Prolift TVM. Ethicon attempted to overturn the verdict but lost again on appeal.
An Indiana court awarded $35 million to a plaintiff alleging injury from Ethicon’s Prolift TVM.
A Philadelphia court awarded $120 million to a plaintiff alleging injury from Ethicon’s TVM. A Pennsylvania court awarded $80 million to a plaintiff alleging injury from Ethicon’s Prolift TVM.
In October 2019, Ethicon agreed to pay $117 million to 41 states and the District of Columbia to settle claims of misleading TVM marketing.
As of March 15th, 2021, Johnson & Johnson has settled the majority of the three federal lawsuits filed in West Virginia. However, the MDL court has remanded several cases back to their local jurisdictions.
Currently, the MDL is closed in the Southern District of Virginia, and plaintiffs can no longer file MDL cases in that court.
Ethicon is currently facing 14,900 TVM lawsuits.

Thousands of individuals have come forward in recent years against the Boy Scouts of America. Specifically, the plaintiffs allege that the Boy Scouts knew that child sexual abuse had been an epidemic within the organization for decades yet failed to do anything about it.
The plaintiffs provided records showing that the organization has kept files of known or accused perpetrators since the 1920s, ensuring these files remained a secret within the organization. These files date back to the 1940s, listing approximately 7,819 perpetrators and 12,254 victims. Most of the perpetrators were Boy Scout leaders who sexually abused victims during camping trips.
Thousands of plaintiffs across multiple states have filed lawsuits against the BSA. The lawsuits allege that the BSA failed to weed out pedophiles, protecting perpetrators rather than its members.
Six men filed a lawsuit against the BSA, claiming that an assistant scoutmaster sexually abused them in the 1980s. The jury was able to read thousands of internal BSA abuse records, making this the first time any plaintiff or jury was allowed to see the mismanagement and cover-ups within the BSA. The case resulted in a $19 million judgment in damages to the six victims.
Investigators reviewed 1,200 secret files from the BSA from 1965 to 1985. Investigators found that authorities in the BSA kept instances of abuse quiet, urging predators to resign rather than reporting them to the police. The LA Times investigated many cases, yet there were no records of BSA officials reporting allegations to the police.
A 20-year-old male filed a lawsuit in California against the BSA for allowing an adult volunteer within the organization to sexually abuse him when he was 13 years old. His attorney won the right to include information at trial from more than 30 years of the “perversion” files the BSA kept as evidence.
The BSA filed for Chapter 11 bankruptcy, listing assets for nearly $10 billion.
In July 2021, the Boy Scouts of America agreed to a landmark $850 million settlement to compensate approximately 60,000 sexual abuse victims under bankruptcy provisions of organizational restructuring. The BSA was supposed to hand over insurance rights to a trust that would oversee claims and compensate the victims. The terms stipulate that the Boy Scouts of America must pay $250 million, with the remaining $600 million to be paid by their 250 local councils. You can find the case here.
A bankruptcy judge set a July 29th, 2021 status hearing regarding the $850 million settlement. At this hearing, U.S. Bankruptcy Judge Laurie Selber Silverstein ruled that the organization that represents more than 70,000 claimants, Abused in Scouting, must provide more details on how the three different plaintiffs’ law firms are involved in the case. The insurers are currently arguing that the group may submit “master ballots” on behalf of individual plaintiffs, which may be inappropriate for the case.
The judge set the next hearing for August 12, 2021. At that hearing, the insurers attempted to block the $850 million deal, stating they were left out of critical settlement negotiations when creating the deal. The insurers argue that the BSA did not use its best business judgment when negotiating the deal.
The judge didn’t address this issue during the hearing, meaning a resolution is still pending.
The Boy Scouts of America are in the process of finalizing the terms of their Chapter 11 bankruptcy reorganization. On July 29, 2022, United States Bankruptcy Judge Laurie Selber Silverstein rejected portions of the proposed bankruptcy plan while approving most aspects of it.
Under the current version of the plan, a $2.7 billion trust fund would be established for the purpose of compensating victims, who would be able to receive an amount between $3,500 and $2.7 million depending on the severity of the abuse experienced.
More than 80,000 men have come forward with sexual abuse allegations against Boy Scouts of America to date.
The opioid epidemic has been widespread throughout the United States since the late 1990s. The medical community believed the pharmaceutical companies when they assured them that patients wouldn’t become addicted to opioid pain relievers. Accordingly, doctors began prescribing opioids at high rates, leading to nationwide addiction, misuse, overdose, and death.
The Centers for Disease Control and Prevention (CDC) reported that opioids have caused quadruple the amount of deaths from 1999 to 2019. They further report that opioids caused more than 70% of 70,630 deaths in 2019.
Opioid lawsuits are widespread, including:
In September 2017, Texas Attorney General Paxton and a bipartisan coalition of 40 additional states served subpoenas and additional requests to eight manufacturers or distributors to investigate their practices in creating and distributing highly addictive painkillers.
On May 15, 2018, Texas filed a lawsuit against Purdue Pharma for deceptive trade practices and misrepresenting the addiction risk of its painkillers, including OxyContin, to doctors and patients.
On March 28th, 2019, New York Attorney General Letitia James filed a lawsuit against Sackler Family and the distributors of opioids for their role in the opioid epidemic.
In 2019, 1,300 city and county entities filed more than 2,400 lawsuits against major opioid manufacturers and distributors. The plaintiffs in these lawsuits accused the companies of creating a public health crisis by mishandling the promotion and dissemination of drugs, causing the deaths of 150 Americans daily and addiction in millions.
They further alleged that the defendants failed to detect, investigate, monitor, and report suspicious subscriptions
of opioids.
There are 50 defendants named in the lawsuits, including these major distributors and their subsidiaries allegedly responsible for 80% of the distribution of opioids:
The lawsuits were assigned to the Northern District of Ohio. The Judicial Panel on Multidistrict Litigation chose this jurisdiction as the most significant location connected to the case. One of the major distributors, Cardinal Health, is based in Ohio, and the state has experienced a spike in opioid overdoses.
A jury found three major pharmacies, Walmart, CVS, and Walgreens, liable for the oversupply of opiates in two Ohio counties on the basis that public nuisance laws were violated. The counties have requested $875 million in damages, according to a Law360 report.
As a result of this bellwether case, lawsuits against pharmacies have gained momentum, on the grounds that the pharmacies erred in dispensing suspicious prescriptions and engaged in deceptive marketing practices.
According to a Reuters report:
Thousands of cases have been transferred to the MDL. In an unprecedented move in April of 2022, Judge Polster ordered that new cases stop being transferred to the MDL because key objectives have already been met and there are few benefits to continuing to add cases. New cases will now be tried in individual courts throughout the U.S.
As of August of 2022, the opioid lawsuits have continued to gain momentum, with both sides experiencing victories. According to a report by NPR, a federal judge rejected the public nuisance argument and ruled in favor of drug company defendants AmerisourceBergen Drug Co, Cardinal Health Inc, and McKesson Corp.
The lawsuit alleged distributors had oversupplied Cabell County and the City of Huntington, West Virginia, but U.S. District Judge David Faber disagreed and stated in the ruling that the companies only distributed drugs to licensed entities and complied with laws requiring suspicious activities to be monitored.
In another ruling reported by U.S. News, the state of West Virginia reached a tentative $161.5 million settlement in May with Teva Pharmaceuticals Inc., AbbVie’s Allergan, and their family of companies in addition to a $26 million settlement in March with Endo Health Solutions.”
The Texas Tribune reported that 52 American states and territories negotiated a $26 billion settlement against McKesson Corp., Cardinal Health and AmerisourceBergen for exacerbating the opioid crisis and overlooking the number of pills pharmacies have been ordering.
The U.S. Court of Appeals for the Sixth Circuit declined a request for a proposed negotiation class to streamline settlement negotiations among over 34,000 municipal entities and opioid manufacturers. A U.S. District Judge previously approved the negotiation class in 2019. However, the Sixth Circuit Court of Appeals denied it on the grounds that it violated federal civil procedural rules.
The Judicial Panel on Multidistrict Litigation ruled to consolidate McKinsey & Co. lawsuits into a separate MDL and transferred the case to the Northern District of California.
The company is battling 17 lawsuits and 22 potentially related causes of action within 11 districts. The panel decided on this move because McKinsey would experience prejudice if they were to join an MDL over three years old.
On February 4, 2021, Texas and McKinsey reached a $38.4 million settlement. The agreement is between 47 states, Washington D.C., five U.S. territories, and McKinsey, resulting in a total settlement of $573 million.
On August 12, 2021, the Plaintiffs’ Executive Committee and the defendants moved for the Court to enter an order approving a Qualified Settlement Fund.

Since 2007, e-cigarettes, or vapes, have become a multibillion-dollar industry in the United States. Over 10.8 million people use JUULs, with one-third of users smoking e-cigarettes daily. E-cigarettes are battery-operated devices that produce heated vapor from a flavored liquid solution that may include nicotine. E-cigarette makers, such as JUUL, market their products as a healthier alternative to cigarettes.
Medical health officials and medical experts have warned consumers of the adverse effects of e-cigarettes. The FDA issued a warning letter to JUUL on September 9, 2019, about their misleading practice of marketing e-cigarettes as safe. They cited multiple false statements from JUUL representatives to students claiming that e-cigarettes were much safer than cigarettes and that the FDA would soon approve them.
According to acting FDA commissioner Dr. Ned Sharpless: “Regardless of where products like e-cigarettes fall on the continuum of tobacco product risk, the law is clear that, before marketing tobacco products for reduced risk, companies must demonstrate with scientific evidence that their specific product does, in fact, pose less risk or is less harmful. JUUL has ignored the law and, very concerningly, has made some of these statements in schools to our nation’s youth.”
North Carolina Attorney General Josh Steil filed a lawsuit against JUUL for “designing, marketing, and selling its e-cigarettes to attract young people and for misrepresenting the potency and danger of nicotine in its products in violation of North Carolina’s Unfair and Deceptive Trade Practices Act.” North Carolina was the first state to initiate legal action against JUUL.
Four school districts in New York, Washington, Kansas, and Missouri sued JUUL. They filed the lawsuits separately, arguing that JUUL created a public nuisance at their schools, intentionally marketed to kids, misrepresented the nicotine content of its products, and endangered teenagers’ health. These lawsuits were part of a coordinated effort by school districts throughout the country. The lawsuits didn’t specify damages and legal fees.
The United States Judicial Panel on Multidistrict Litigation transferred cases against JUUL to the Northern District of California before Judge William H. Orrick.
In June 2021, JUUL settled a $40 million lawsuit filed against them by North Carolina for marketing e-cigarettes to minors. This settlement was their first with a state. They agreed to pay the settlement over six years and to change their nationwide marketing targeting minors. JUUL didn’t admit any wrongdoing as part of the settlement and stated that the agreement was in line with their ongoing efforts to combat underage vaping and reduce harm in adult smokers.
In July 2021, U.S. District Judge William Orrick in San Francisco ruled that current and former directors of JUUL, and Altria (its largest investor), couldn’t dismiss 18 bellwether cases in the MDL pending against the e-cigarette manufacturer. However, the Judge did dismiss strict liability claims against the individual defendants and some state law claims.
The FDA banned the continued marketing of Juul products on June 23, 2022, but this was stayed by the U.S. Court of Appeals for the D.C. Circuit the following day at the company’s request pending the court’s review of Juul’s case. On July 5, 2022, the FDA administratively stayed the marketing denial order on the grounds that the scientific issues warrant further review. The FDA noted that this is a temporary suspension of the ban, not a rescission.
The original reason for the FDA ban was due to insufficient safety evidence and conflicting information in the company’s premarket tobacco product applications regarding the genotoxicity and potentially harmful chemicals that leach from JUUL’s e-liquid pods.
School districts have taken center stage in the fight against the vaping company. More than 700 school districts across the country have filed lawsuits against Juul as of March of 2022, according to ClarksvilleNOW, a local news affiliate in Clarksville, Tennessee.
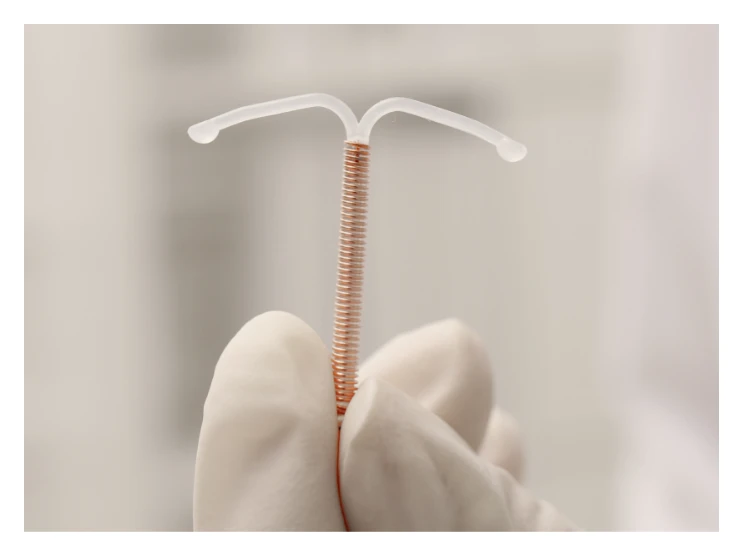
Intrauterine devices (IUDs) have been a form of long-term contraception on the market for more than 30 years. They’re hormone-free T-shaped copper devices inserted into the uterus to provide immediate pregnancy protection.
Experts considered Paragard IUDs one of the safest forms of contraception as they are surgery-free, hormone-free, and inserted in-office.
Lawsuits filed by individuals injured by Paragard IUDs include:
Stephanie Ideus and other plaintiffs filed lawsuits against Teva Pharmaceuticals in 2016. These injured parties claimed that the Paragard IUD broke and embedded in their uteruses during removal, requiring surgery. In February 2019, The Eighth Circuit Court of Appeals stated that under Nebraska’s Learned Intermediary Law, Teva Pharmaceuticals was not required to warn Paragard IUD users directly of the risk of breakage but only had a duty to warn physicians, and the company complied accordingly.
On September 11, 2020, Georgia Bowers filed a complaint against Teva Pharmaceuticals and various other defendants. Both parties demanded a jury trial, and the case was transferred to the U.S. District Court for the Northern District of Georgia before Leigh Martin May. Bowers filed the lawsuit after broken pieces of the Paragard IUD remained in her body after removal by her doctor. As a result, she had to undergo a hysteroscopy to remove the remaining piece of the IUD.
Carley Tredway had her Paragard IUD implanted in 2008. In 2018, her doctor removed the device, leading to injuries. As a result, she filed a lawsuit against Teva Pharmaceuticals, seeking compensation and punitive damages.
In most of these cases, the plaintiffs claim that they lacked information about the dangers of the Paragard IUD, specifically that it could potentially break and become embedded in the uterine wall, causing damage to the uterus and other organs. Paragard IUD lists device breakage as a potential side effect and warns that they had previously received reports of the possibility of breakage of an embedded Paragard IUD during non-surgical removal. Still, the frequency of such occurrence was not readily available as the company failed to disclose that information.
Primary allegations in the lawsuit include:
Manufacturing and Design Defects: Focusing on the risk of Paragard IUD breaking inside the uterus during ordinary use and removal
Failure to Warn: Asserting that Teva Pharmaceuticals, manufacturers, and distributors of Paragard IUD failed in their duty to adequately warn consumers of the risk of breakage (plaintiffs claim that although their doctors may have understood the risks, patients were unaware, thereby violating their right of informed consent)
On December 16, 2020, the Judicial Panel on Multidistrict Litigation ordered 55 Paragard IUD lawsuits pending in 31 districts to be consolidated and assigned to the Northern District of Georgia. Twenty-five additional cases are pending consolidation.
Teva Pharmaceuticals claimed that the variation in the cases among plaintiffs made them unsuitable for consolidation, but the court rejected the defendant’s claims citing that the plaintiffs’ cases involve “common questions of fact” regarding Paragard IUD’s propensity to break upon removal.
As of August of 2022, the FDA Adverse Events Reporting System (FAERS) reported 45,382 adverse events associated with the Paragard device. On July 15, 2022, there were 1,246 pending cases in the MDL, which was consolidated in the U.S. District Court of Georgia in December of 2020. Cases are still being accepted, but no bellwether trials are scheduled.
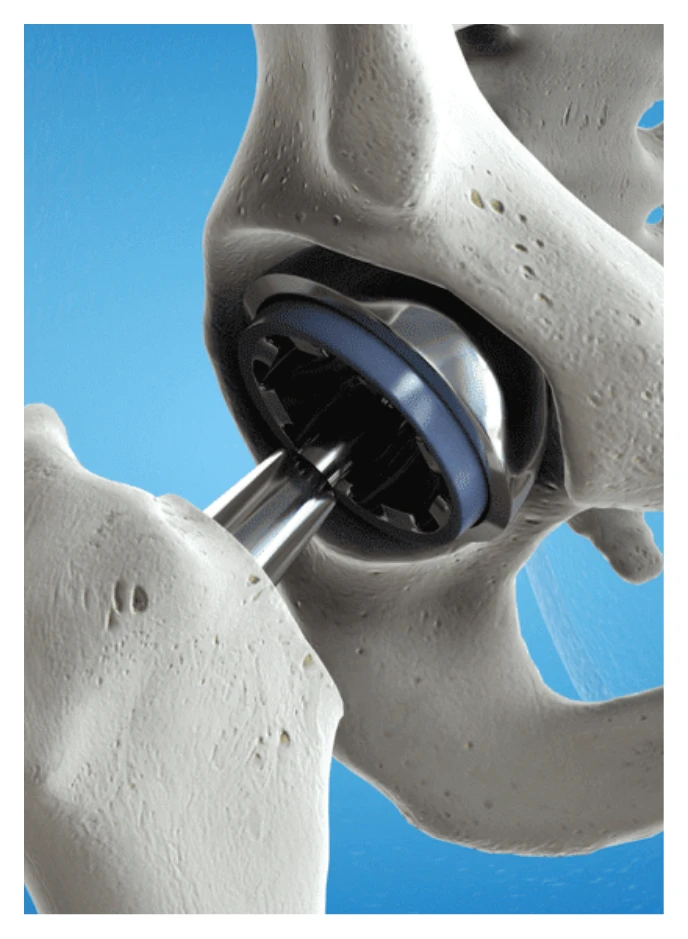
In 2012, the FDA’s Orthopedic and Rehabilitation Devices Advisory panel reviewed the failure rates, complications, and risk factors associated with artificial hip replacements. After this review, in 2013, the USDA modified its artificial hip regulations.
This modification led to the reduction in the use of metal-on-metal hip implants. The hip replacements had a high failure rate due to the implant loosening, causing:
After recalling its product in 2010, DePuy, a subsidiary of Johnson & Johnson and the DuPuy Pinnacle hip implants manufacturer, established a program in 2013 to resolve issues involving their hip implants.
Smith & Nephew is another hip implant maker using metal liners in its products. The medical device manufacturer recalled 2,000 devices in 2016, three years after becoming aware of the problem. The company has paid out more than $40 million in settlements, and 765 cases are still pending in a consolidated MDL in the U.S. District Court for the District of Maryland.
On Feb. 28, 2022, Law360 reported that U.S. District Judge Catherine C. Blake dismissed 175 claims involving male patients who received 50-millimeter or larger femoral heads. According to the ruling, only their claims for misrepresentation and breach of express warranty may move forward.
This order was issued in response to Smith & Nephew’s request for summary judgment with regard to these grounds and failure to warn the FDA, negligence, failure to train and misbranding, and punitive damages.
The court ruled in Smith & Nephew’s favor on all except the misrepresentation and warranty claims, citing the need to decide each case individually.
In December of 2021, Smith & Nephew filed a motion to dismiss claims related to 2009 implants in the larger MDL on the basis that plaintiffs failed to show that the company had information about defects that would affect their use before then. A ruling is still pending.
Lawsuits filed by injured hip replacement recipients include:
Trials began against DePuy in the United States District Court Northern District of Texas under MDL 3:11-md-02244.
A Dallas jury awarded five plaintiffs a $502 million verdict against Johnson & Johnson for their use of defective DePuy Pinnacle hip implants and failure to warn patients of their possible risks. The jury calculated the award at $142 million in compensatory damages and $360 million in punitive damages intended to punish the defendant for their egregious actions and deter future wrongdoing.
Pennsylvania Attorney General Josh Shapiro and 46 other attorney generals reached a $120 million agreement or Consent Judgment with DePuy and Johnson & Johnson DePuy. The lawsuit alleged that DePuy engaged in deceptive sales practices and exaggerated the longevity claims for their metal-on-metal hip replacement devices.
587 lawsuits filed against Smith & Nephew’s Birmingham Hip Resurfacing (BHR) regarding their defective hip implants were pending in multidistrict litigation in the United States District Court for the District of Maryland.
Exactech recalled the Connexion GXL polyethylene acetabular hip implant liners on June 29, 2021 due to a risk of excessive edge loading and premature wear of the polyethylene material, especially with regard to its thinnest products. Hip implants should last 20 years, according to the American Academy of Orthopaedic Surgeons, but the Exactech implants may wear in as little as three to six years.
Hip implants are not the only joint replacement Exactech has recalled. Exactech also recalled knee and ankle implants on August 31, 2021 due to premature wear of the polyethylene inserts. This was brought on by improper packaging that has allowed oxygen permeation of the insert, which decreases the life of the implant.
The result is that an unusually high percentage of patients are being injured by particles from the worn inserts entering surrounding tissues, leading to infection, osteolysis, and
other injuries.
This necessitates revision surgery, which is painful, more complicated, and often contraindicated for elderly patients with other health conditions.
One of the more common complications, osteolysis often presents without symptoms, resulting in advanced bone loss by the time the condition is discovered. This can lead to failure of the device and, in the most severe cases, the necessity of amputation above the knee.
The number of cases is growing around the country, and they are expected to be transferred to multidistrict litigation. Exactech is primarily communicating with physicians about the recall, so strong advertising by plaintiffs’ attorneys will be necessary to make the public aware of the possibility of having received defective implants.

Research from as early as the 1970s suggests a link between the use of talcum powder and the development of ovarian cancer. Johnson & Johnson, a household name for talcum powder, has been manufacturing and distributing the widely used consumer product for decades.
However, in 2018, Reuters released a report exposing Johnson & Johnson for knowing that asbestos was in its baby powder products. The lawsuits claim that the manufacturers knew that talc contained asbestos, which can cause ovarian cancer and mesothelioma. Yet, despite knowing the risks, these companies didn’t do anything to warn consumers about these risks. While many individuals filed their own lawsuits, there are at least four class action or mass lawsuits.
The first plaintiff filed a lawsuit in the Federal District Court in South Dakota. A jury found that Johnson & Johnson was negligent but didn’t award damages or compensation to the plaintiff, Deane Berg.
4,800 plaintiffs filed lawsuits against Johnson & Johnson. The plaintiffs alleged that Johnson & Johnson failed to heed warnings from multiple studies of the talc-based powder’s carcinogenic (cancer-causing) effects. As a result, plaintiffs asserted that the use of Johnson & Johnson’s talcum powder caused them to develop ovarian cancer.
On August 21, 2017, a Los Angeles jury awarded $417 million in compensatory and punitive damages to Eva Echevarria, a 63-year-old plaintiff. She had used Johnson & Johnson talcum powder since she was 11 years old and was diagnosed with ovarian cancer in 2007. However, the ruling was overturned on October 20, 2020, citing a lack of sufficient expert testimony on whether there’s specific causation linking talcum powder use to the plaintiff’s ovarian cancer.
Nearly 20,000 cases were consolidated into an MDL in the U.S. District of New Jersey. Johnson & Johnson agreed to settle 1,000 of these cases for $100 million. The MDL is currently proceeding in the U.S. District Court of New Jersey, with plaintiffs alleging that regular genital application of baby powder and shower-to-shower talcum powders contributed to the development of ovarian cancer.
On April 27, 2020, a ruling was rendered in the MDL, allowing plaintiffs’ expert witnesses to testify that Johnson & Johnson’s baby powder contains asbestos and talc powder can cause ovarian cancer.
On October 19, 2020, a California jury awarded Plaintiff Philip Devonian an $18.07 million verdict after discovering that repeated exposure to the asbestos particles in Johnson & Johnson talcum powder caused his advanced mesothelioma, a fatal type of lung cancer.
The National Council of Negro Women filed a lawsuit against Johnson & Johnson, alleging the company of “knowingly deceptive marketing to Black women” for decades. They argue that the company provided free samples at beauty salons, had radio campaigns, and other efforts.
As of July 18, 2021, Johnson & Johnson is exploring creating a new business that would then seek bankruptcy protection after Johnson & Johnson offloads its liabilities from its baby powder litigation. If they pursue a bankruptcy plan, plaintiffs would likely receive lower payouts if they’re unable to settle before the pharmaceutical company’s declaration.
In October of 2021, in a move known as the “Texas two-step,” Johnson & Johnson transferred its liabilities to a new subsidiary in Texas, LTL, then filed for bankruptcy in North Carolina, while keeping its own assets separate, according to a report by NPR.
Johnson &Johnson has offered to create a fund worth billions through the bankruptcy court for the benefit of cancer victims, which would allow victims to file claims without going through the court system. This can help victims receive compensation faster, but the settlements could be lower. The company still has 38,000 unresolved cases.
According to a Forbes report, Johnson & Johnson has set aside $3.9 billion in anticipation of the establishment of the trust fund.

Proton pump inhibitors are drugs formulated to regulate acid production to treat heartburn, gastroesophageal reflux disease (GERD), esophageal inflammation, and small intestine and stomach ulcers.
Approximately 15 million Americans use proton pump inhibitors. However, multiple studies published in the Journal of the American Medical Association and the British Medical Journal discovered that proton pump inhibitors caused adverse health effects, including:
More than 15,000 people have filed lawsuits against the following manufacturers of proton pump inhibitors:
The plaintiffs argue that the manufacturers failed to warn consumers and medical professionals about the risks associated with their products. Some Prilosec plaintiffs claim that AstraZeneca knew the risks associated with their proton pump inhibitors for at least ten years.
People injured after taking proton pump inhibitors filed the following lawsuits:
Harry Mason filed a lawsuit in the Southern District of Illinois against AstraZeneca after using proton pump inhibitors since 2006. He alleged that the use of the drug led to his kidney failure and transplant.
Penelope Costamagna filed a lawsuit in 2017, alleging that she required a kidney transplant after taking Prilosec for multiple years. There was a high likelihood that she would need a second life-saving kidney transplant.
Jeanette Stockton filed a lawsuit in 2017, alleging that after using Nexium from 2002–2012, she was diagnosed with chronic kidney disease in 2012 but continued to take Nexium until 2016, not knowing that it was the cause of her kidney disease.
On August 2, 2017, various proton pump inhibitors cases were pending throughout the country.
The U.S. Judicial Panel on Multidistrict Litigation consolidated the cases in the District of New Jersey.
In addition, the presiding judge ordered defendants and plaintiffs to select 20 representative cases for a discovery pool to be condensed to six trials, with the first trial scheduled to commence on November 15th, 2021.
The Bellwether trial schedule for the first three trials has been set, and the cases are scheduled to be heard as follows:
The judge dismissed the defendant’s Daubert motions on July 19, 2022. The defendants had requested a dismissal based on preemption, but (page 6 of downloaded document) under Ohio law, which governs in this case, the companies would have to show that the doctor would not have prescribed differently had they been warned. The doctors’ testimony was not sufficient to prove this.
Valsartan is the active ingredient in the blood pressure and heart failure medication sold under the brand name Diovan. Approximately three million Americans use this medication annually. In 2018, the Food and Drug Administration (FDA) discovered that several drug products that contained valsartan might have been contaminated with carcinogens called N-nitrosodimethylamine (NDMA) and N-Nitrosodiethylamine (NDEA). These impurities can cause multiple forms of cancer, including liver, stomach, colon, rectum, and pancreatic. The discovery prompted an FDA nationwide recall of 600 batches of valsartan.
In February 2019, the U.S. Judicial Panel on Multidistrict Litigation consolidated valsartan lawsuits in the New Jersey federal court. The plaintiffs name the following defendants in their lawsuits:
The 10 initial lawsuits sought six years’ worth of reimbursement for plaintiffs that had purchased contaminated batches of valsartan. However, the court acknowledged 17 additional personal injury lawsuits for plaintiffs that had developed cancer or liver damage from using valsartan.
The parties are currently in discovery proceedings. The most recent order allowed an extension for plaintiffs to file their motion to compel no later than July 23, 2021. Defendants, ZHP, had until August 13, 2021, to file their opposition. As of July 20, 2021, the U.S. Judicial Panel on Multidistrict Litigation continues to add lawsuits to the MDL, with just over 900 cases currently pending as of August 2021.
Mass tort cases will never cease to exist. Pharmaceutical companies are notorious for distributing and marketing drugs that cause injuries, prioritizing profit over public health.
Besides pharmaceutical companies, many businesses disregard their duty to safeguard the public’s well-being to pursue increased revenues, creating the need for lawsuits to hold these companies liable.
Below is information on several notable ongoing mass tort cases to watch in 2022 and beyond. These cases are constantly developing, and the information will be updated as they proceed:
Necrotizing enterocolitis (NEC) is a deadly illness that most often impacts premature infants, according to Advances in Nutrition. A strong link between the use of bovine-based infant formulas and NEC has been established, with breastmilk being established as a significant protective factor.
Yet Abbott Laboratories, the maker of multiple formula brands, has engaged in a longstanding practice of marketing bovine-based formulas as an equal substitute for breastmilk while failing to mention the risk of developing NEC.
There are currently 106 pending actions in MDL 3026 in the Northern District of Illinois.
Belviq, manufactured by Arena Pharmaceuticals and distributed by Eisai Inc., is a weight loss drug prescribed for weight management of health conditions, including diabetes and high blood pressure. Eisai conducted an FDA-mandated five-year double-blind study with over 12,000 participants. The study showed that Belviq usage increased the risk of pancreatic, colorectal, and lung cancer, prompting an FDA recall of Belviq nationwide in February 2020.
Plaintiffs in seven actions moved to consolidate the litigation into an MDL in the Eastern District of Louisiana on April 12, 2020. The defendants’ attorneys opposed the motion on May 5, 2021, claiming that the cases aren’t similar enough to warrant consolidation. On August 10, 2021, the U.S. Judicial Panel on Multidistrict Litigation denied the transfer, stating that the 13 actions before them, including 12 individual personal injury actions and one putative class action, aren’t enough actions to warrant an MDL. The Panel said that while the number of cases might grow, presently, there are only 20 actions, at most.
PFAS is an acronym used for a group of manufactured chemicals, including perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS). Often, people call them “forever chemicals” as they’re not biodegradable and can remain in the human body for decades. The dangers of PFAS chemicals became more widely known in the early 2000s when chemical manufacturer DuPont faced scrutiny for contaminating the drinking water supply of 70,000 West Virginia residents.
Studies link ingesting PFAS chemicals to health problems, including thyroid disease, ulcerative colitis, leukemia, and cancers of the kidneys, liver, testes, and pancreas. MDL-2873 currently has approximately 1,450 cases pending before the United States District Court District of South Carolina, under In Re: Aqueous Film-Forming Foams Products Liability Litigation.
More than 700 military bases have also sued DuPont, 3M, and other companies that manufacture the firefighting foam used in military training drills. The bases allege that the companies knew their products contained PFAS chemicals that could contaminate local groundwater but failed to take measures to protect the public.
A complete list of defendants named in the lawsuits follows:
In July 2021, a court awarded Delaware a $50 million settlement with three prominent local chemical manufacturers DuPont, Chemours, and Corteva. The companies have also agreed to pay an additional $25 million to the state if similar claims are settled in other states exceeding $50 million.
New York-based artificial intelligence company Clearview AI founded in 2017, developed facial recognition software targeted for law enforcement agencies.
The technology gathered publicly available images from multiple social media platforms such as Facebook, Twitter, YouTube, and Instagram.
The New York Times published an article in January 2020 claiming that Clearview AI’s algorithm had gathered more than 3 billion images and stored them in their database.
This article prompted a lawsuit from the American Civil Liberties Union (ACLU) against Clearwater AI for violating the Illinois Biometric Information Privacy Act (BIPA). As a result, the Northern District approved an MDL in December 2020.
Plaintiffs filing lawsuits demanded that Clearview AI put proper measures in place to protect the existing sensitive biometric data from hackers and other unlawful uses or distribution. They also requested an injunction to stop the company from selling Illinois residents’ biometric data and immediately delete all collected information.
At a hearing held on May 25, 2021, Clearview AI asked that presiding Judge Sharon Johnson Coleman dismiss the lawsuit, claiming that their collection and dissemination of such is protected under the First Amendment and the Illinois Constitution. Clearview AI further claimed that the plaintiffs failed to prove their data was collected in Illinois and that most data collection occurred outside Illinois, which is grounds for dismissal.
The most recent court proceeding was a status hearing on May 27, 2021.
Camp Lejeune is a Marine Corps military base in Jacksonville, North Carolina, where military members trained and resided with their families. From August 1, 1953 through December 31, 1987, the water at Camp Lejeune was contaminated with volatile organic compounds (VOCs), which included PCE, TCE, vinyl chloride, and benzene.
The CDC has linked these VOCs to severe and deadly health effects, including cancer, birth defects, childhood leukemia, Parkinson’s disease, kidney disease, neurological effects, and immune system impairment. The military discovered the contamination at least as early as 1982 and closed down the most contaminated wells in 1985.
Although the military notified Camp Lejeune military members living on the site at the time the contamination was addressed, former Camp Lejeune residents were not notified until 1999, according to a report by North Carolina Health News. The State of North Carolina has a ten-year statute of repose that time-barred water contamination victims from filing a civil lawsuit for damages.
Jerry Ensminger resided on the base with his family during the contamination. His daughter Janey died of leukemia in 1985 at the age of nine. When he learned of the contamination in 1999, he became a lead advocate for reform.
Thanks to his efforts, the Janey Ensminger Act became law in 2012, which gave veterans the right to obtain service-connected VA benefits for Camp Lejeune water contamination, which included reimbursement for affected family members’ medical expenses. However, this legislation stopped short of providing victims with civil relief.
In August of 2022, the Camp Lejeune Justice Act, which is part of the Honoring Our PACT Act, became law. The new law sets aside the North Carolina statute of repose in cases involving victims of Camp Lejeune water contamination and allows them to file civil lawsuits against the military for compensatory damages.
The Agency for Toxic Substances and Disease Registry estimates that over a million military personnel, civilian employees, and their families were impacted, and the number of lawsuits and amounts of compensation awarded are expected to be substantial.
Philips CPAP machines were voluntarily recalled due to a defect in the soundproofing foam contained within the devices. This foam, when inhaled, carries severe health risks. The defect has impacted a significant number of users resulting in a significant number of lawsuits. As a result, the case was consolidated to multidistrict litigation and transferred to the Western District of Pennsylvania on October 8, 2021.
Guideposts Solutions, an independent investigative agency, has uncovered decades of widespread sexual abuse and violence in Southern Baptist churches, which was subsequently concealed by high-ranking officials and their lawyers. The 288-page report outlines the shocking maltreatment survivors received when coming forward.
Now that this information has been made public, survivors can finally hold the Southern Baptist Convention accountable for its role in aiding and abetting the abuse while further traumatizing survivors.
Mass tort cases are time-consuming and require ample financial resources. In addition to actual litigation work, attorneys lose time on administrative tasks that involve large volumes of data that must be gathered, reviewed, and managed.
For these reasons, outsourcing case management is crucial. Legal outsourcing companies can provide highly competent staff specialized in multiple areas of litigation at reduced costs.